Blood testing is one of the most essential tools in modern healthcare. Behind every accurate test result lies a carefully preserved blood sample. That preservation depends heavily on anticoagulants—special substances added to blood collection tubes to prevent clotting. Among the most widely used additives are EDTA and Sodium Citrate, two anticoagulants that serve very different purposes in laboratory medicine.
Understanding the difference between EDTA and Sodium Citrate is critical for laboratories, clinics, distributors, and healthcare professionals who rely on consistent diagnostic accuracy. Choosing the wrong tube can affect results, delay diagnosis, and increase laboratory costs.
In this comprehensive guide, we explore how these anticoagulants work, where they are used, and how to choose the correct tube for your clinical or laboratory workflow.
For an overview of professional-grade blood collection supplies, you can explore the official edtatube resource, which provides detailed technical insights and product information.
What Is EDTA and How Does It Work?
EDTA, short for Ethylenediaminetetraacetic Acid, is one of the most widely used anticoagulants in laboratory medicine. It is particularly valued for its ability to preserve blood cell structure during analysis.
According to the detailed explanation of EDTA chemistry on Wikipedia (wiki/Ethylenediaminetetraacetic_acid), EDTA functions by binding calcium ions, which are essential for blood clotting. Without calcium, the coagulation cascade cannot proceed.
How EDTA Prevents Blood Clotting
EDTA works through a process called chelation, which involves tightly binding calcium ions in the blood. Calcium plays a central role in activating clotting factors. When EDTA binds calcium:
- The clotting cascade stops immediately
- Blood remains fluid
- Cellular structure stays intact
- Platelet aggregation is prevented
For a deeper understanding of this mechanism, refer to how-edta-anticoagulant-works-in-blood-collection-tubes, which explains the process in practical laboratory terms.
Common Uses of EDTA Tubes
EDTA tubes are widely used in hematology because they maintain the shape and integrity of blood cells.
Typical applications include:
- Complete Blood Count (CBC)
- Hemoglobin testing
- White Blood Cell differential count
- Platelet count
- Blood typing
- Reticulocyte analysis
These tests require intact cells, making EDTA the gold standard anticoagulant for hematology.
Healthcare providers frequently rely on professional-grade products such as those listed at:product-category/edta-tube
What Is Sodium Citrate and How Does It Work?
Sodium Citrate is another widely used anticoagulant, but it operates differently from EDTA. Unlike EDTA, which permanently binds calcium, sodium citrate binds calcium temporarily, allowing clotting to be reversed during laboratory testing.
This reversible property makes sodium citrate the preferred choice for coagulation studies.
How Sodium Citrate Prevents Clotting
Sodium citrate prevents clotting by binding calcium ions loosely. This temporary binding allows laboratory technicians to add calcium back into the sample when needed.
This feature enables controlled clot formation during testing.
Typical mechanism:
- Sodium citrate binds calcium
- Clotting temporarily stops
- Laboratory adds calcium later
- Controlled clotting begins
Because of this reversible mechanism, sodium citrate is essential in coagulation analysis.
Applications of Sodium Citrate Tubes
Sodium citrate tubes are commonly recognized by their light blue caps and are mainly used for coagulation studies.
These include:
- Prothrombin Time (PT)
- Activated Partial Thromboplastin Time (APTT)
- D-dimer testing
- Fibrinogen testing
- Coagulation factor analysis
These tests require precise clotting control, which sodium citrate enables.
EDTA and Sodium Citrate: Side-by-Side Comparison
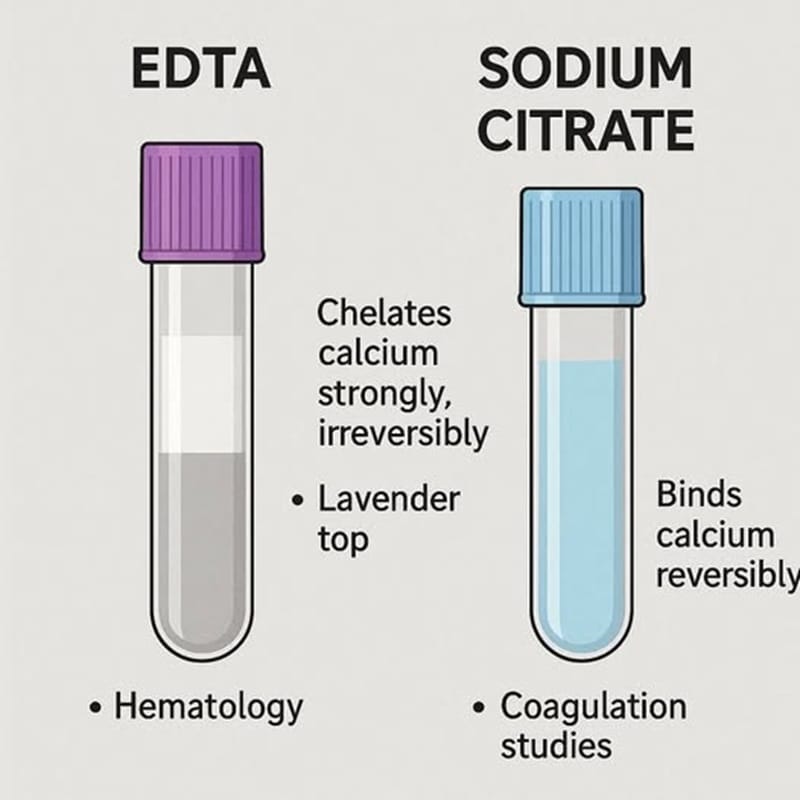
Understanding the difference between EDTA and Sodium Citrate becomes easier when their characteristics are compared directly.
Comparison Table: EDTA vs Sodium Citrate
| Feature | EDTA | Sodium Citrate |
|---|---|---|
| Anticoagulant Type | Permanent calcium binding | Temporary calcium binding |
| Mechanism | Chelation | Reversible binding |
| Primary Use | Hematology | Coagulation testing |
| Tube Color | Purple / Lavender | Light Blue |
| Blood Ratio Sensitivity | Moderate | Very High |
| Cell Preservation | Excellent | Moderate |
| Coagulation Testing | Not Suitable | Required |
This comparison highlights why choosing the correct anticoagulant is critical for accurate diagnostics.
You can review additional product details here:products
Why EDTA Is Preferred for Hematology
Hematology focuses on analyzing blood cells. Therefore, preserving cell structure is essential.
EDTA excels in this role because it:
- Maintains red blood cell shape
- Prevents platelet clumping
- Preserves white blood cells
- Prevents clot formation quickly
Because of these advantages, EDTA tubes—especially purple-cap tubes—are widely used worldwide.
More detailed product information is available here:siny-edta-anticoagulant-tube-purple-cap
Why Sodium Citrate Is Preferred for Coagulation Testing
Coagulation testing requires blood clotting to occur under controlled laboratory conditions.
Sodium citrate enables this by:
- Allowing clotting to restart
- Maintaining test accuracy
- Supporting coagulation factor testing
For tests like PT and APTT, even small changes in sample volume can affect results. That is why sodium citrate tubes must maintain a precise 9:1 blood-to-anticoagulant ratio.
How to Choose Between EDTA and Sodium Citrate Tubes
Selecting the correct anticoagulant tube depends mainly on the type of test being performed.
Laboratories should consider:
- Type of analysis
- Required accuracy level
- Sample stability
- Instrument compatibility
- Regulatory compliance
For hematology testing, EDTA tubes remain the top choice.
For coagulation testing, sodium citrate tubes are essential.
Detailed product specifications can be found at:edta-tubes-for-blood-collection
Importance of Blood-to-Anticoagulant Ratio
One of the most critical differences between EDTA and Sodium Citrate involves sample ratio sensitivity.
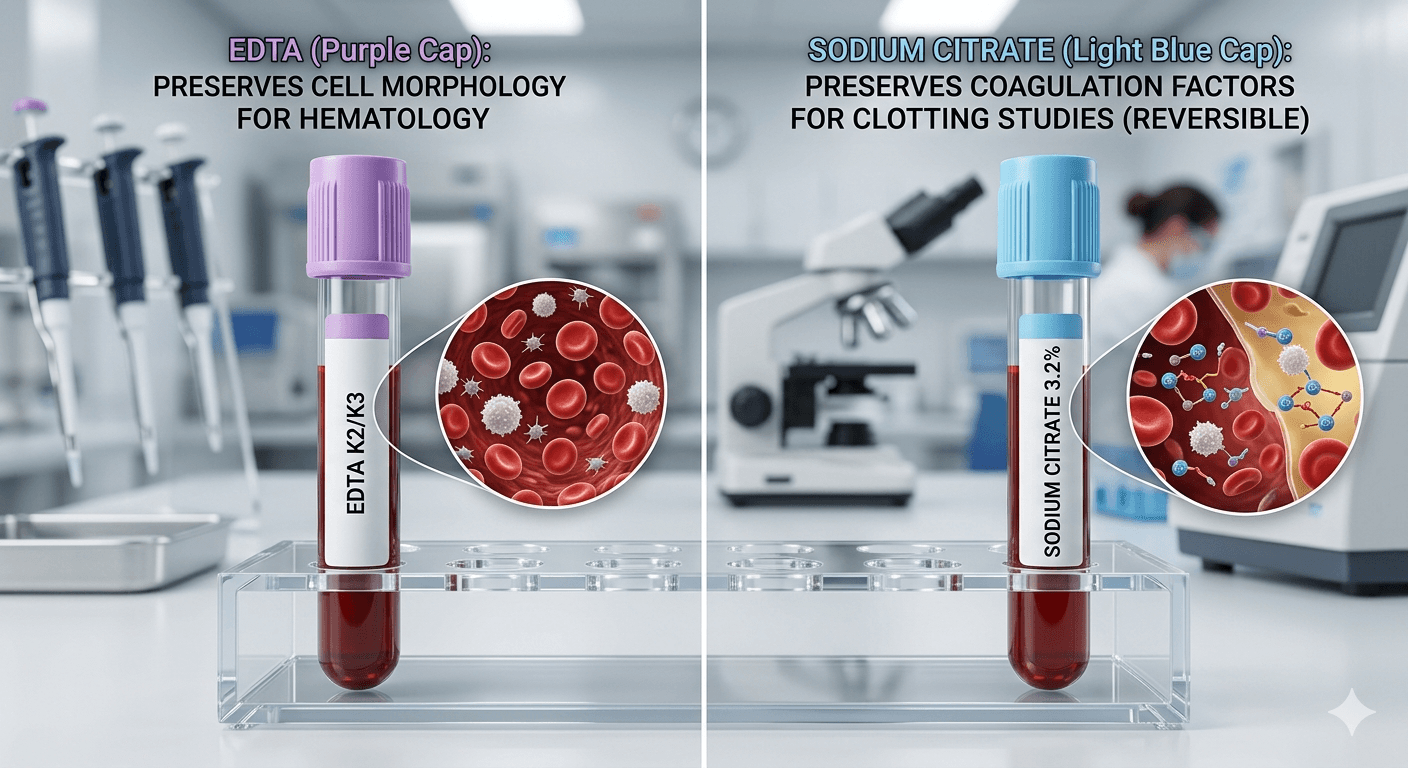
EDTA tubes tolerate minor filling variations. However, sodium citrate tubes require exact filling levels.
Incorrect ratios may lead to:
- False test results
- Sample rejection
- Delayed diagnosis
Laboratories often rely on automated filling systems to maintain consistency.
Manufacturing Quality Matters in Blood Collection Tubes
The reliability of anticoagulants depends heavily on manufacturing quality.
High-quality tubes provide:
- Consistent vacuum pressure
- Uniform additive coating
- Leak-proof design
- Sterile packaging
Working with trusted suppliers helps reduce laboratory errors.
For additional manufacturing insights, visit:edta-tube
You can also explore laboratory product listings here:sinymedical.en.made-in-china.com
Educational video resources are available through:youtube.com/@sinymedical
Global Demand for EDTA and Sodium Citrate Tubes
The demand for anticoagulant tubes continues to rise due to increased laboratory testing worldwide.
Factors driving growth include:
- Aging populations
- Chronic disease prevalence
- Expanded diagnostic testing
- Technological advancements
According to industry reports, the global blood collection tube market continues to grow steadily each year.
Reliable suppliers such as those listed atcontact-us
support laboratories with consistent supply chains.
Summary and Final Thoughts
The decision between using EDTA and sodium citrate is dictated by the biological requirements of the test. EDTA is the robust, reliable choice for looking at the cells themselves—counting them, measuring them, and checking their health. Sodium citrate is the delicate, precise choice for looking at the liquid “machinery” of the blood and how it reacts to injury.
By ensuring your facility uses the highest quality tubes and following the correct clinical protocols, you guarantee that every drop of blood collected provides a clear, accurate window into the patient’s health. For more detailed comparisons on which additive is right for your specific needs, check out our comprehensive guide on EDTA vs Citrate tubes: the best anticoagulant.
FAQs
Q. Which anticoagulant is better for a Complete Blood Count (CBC)?
A. EDTA is significantly better for a CBC. Because EDTA prevents platelet clumping and preserves the shape of red and white blood cells for a long period, it ensures that the automated counters provide the most accurate results for EDTA and sodium citrate comparisons in hematology.
Q. Can I use a sodium citrate tube if I run out of EDTA tubes?
A. No, you generally cannot. Sodium citrate dilutes the blood (due to its liquid state and 9:1 ratio), which would result in artificially low cell counts. To understand the specific chemical reasons why this doesn’t work, you can refer to the Wikipedia page on EDTA.
3. Why is the 9:1 ratio so important for sodium citrate?
A. The 9:1 ratio is critical because coagulation tests are designed based on a specific concentration of citrate. If there is too much or too little blood relative to the EDTA and sodium citrate balance required for the reagents, the clotting times (PT/PTT) will be inaccurate, potentially leading to incorrect medication dosing for the patient.
Q. Are there different types of EDTA?
A. Yes, the most common are $K_2EDTA$ (dipotassium) and $K_3EDTA$ (tripotassium). $K_2EDTA$ is generally preferred because it is spray-dried and has less effect on cell volume. You can find both varieties in our products section.
Q. How long is a sample stable in EDTA and sodium citrate?
A. Samples in EDTA are usually stable for CBC for up to 24 hours at room temperature, though blood smears should be made sooner. Sodium citrate samples for coagulation are much more time-sensitive and should often be tested within 2 to 4 hours to ensure the clotting factors don’t degrade.